This blog is currently paused. We hope to secure more research funding to continue the project and start writing again. For now, take a look at our substack.
Enslaved American Silversmiths?
The use of slaves in making has attracted careful attention from scholars of North American material culture, for example Catherine Bishir’s Crafting Lives (2013), and Glenn Adamson’s Craft: An American History (2021). We also have decades of fine scholarship on the use of enslaved West African ironworkers in colonial North America. The importance of enslaved blacksmiths is well established and reflected in major museum collections. The brutal lives and deaths of slaves at iron foundries and iron ore mines have been documented, including their exposure to heavy metal toxins. However, there is comparatively little written about workers of non-ferrous metals.[1]
In Bishir’s detailed study of New Bern, NC, the silversmiths were all free, including the literally named Freeman Woods. Adamson paints on a far broader canvas. He introduces us to Native American silversmiths, such as Slender Maker of Silver, and reminds us that “When we are faced with an 18th-century teapot or high chest in a museum, it is usually impossible to know whether or not it was made partly with enslaved labor.” Adamson is certainly referring to a silver teapot: He begins his book with a portrait of Paul Revere (Boston MFA 30.781), painted in 1768 holding in his left hand a silver teapot that he has crafted, not unlike Agatho. Like Agatho, too, Revere is shown with the tools of his trade, with which he is about to decorate and engrave the teapot.[2]
A coffeepot crafted by Revere just a year after Copley painted his portrait is in the Yale University Art Gallery (2016.158.1).[3] Paul Revere learnt his craft from his father, Apollos Rivoire (later Paul Revere Sr.), who had been an indentured servant as an apprentice to John Coney. The Reveres owned no slaves. This also appears to be the case with other silversmiths of the period, for example Richard van Dyck (1717-70) and Myers Myers (1723-95), who worked in New York City. The 1703 census is the only record that silversmiths in New York owned slaves. Three of thirteen silversmiths at that time each owned a single female slave, almost certainly a housekeeper. Otherwise, there is no evidence that any New York silversmith ever used slaves in their workshops.[4]
Many silversmiths owned slaves. For example, the probate inventory of Charleston silversmith Thomas You (d. 1786) lists seventeen slaves, but none is said to have been a smith.[5] I am aware only very few instances where it is explicitly stated that slaves were trained silversmiths or goldsmiths (the terms are used interchangeably). One is Abraham, who was owned and trained by the Charleston silversmith Alexander Petrie (d. 1768).[6] It is possible that further information may be found in the account book of Austen and Laurens, slave traders in Charleston, now at Yale.[7] Newport, a goldsmith from Newport, RI, owned and trained by Isaac Anthony, was reported as a runaway in 1749. Four years later, another Newport smith, advertised in a Boston newspaper that he wishes to sell an unnamed goldsmith, possibly the same Newport, but probably not.[8]
[1] K. W. Bruwelheide et al., “Restoring identity to people and place: Reanalysis of human skeletal remains from a cemetery at Catoctin Furnace, Maryland,” Historical Archaeology 54 (2020), 110-37.
[2] C. W. Bishir, Crafting Lives. African American artisans in North Bern, North Carolina (Chapel Hill, NC: UNC Press, 2013); G. Adamson, Craft: An American History (London and New York: Bloomsbury, 2021). For the portrait of Revere, by John Singleton Copley, see: https://collections.mfa.org/objects/32401
[3] Yale Art Gallery has many pieces of silver fashioned by Revere, including a silver teapot made in 1795 (1930-959).
[4] K. Hillen McKinsey, New York City Silversmiths and their Patrons, 1687-1750, Unpublished dissertation, University of Delaware 1984. We cannot be so certain that they did not have indentured servants – workers who were contracted to provide unpaid labor for a period of time, usually five to seven years, in payment of a debt – since this form of servitude was so very common among early immigrants. Debt bondage was usually related to the cost of their passage to North America.
[5] H. Nowell Brazier, “The most elegant and general assortment of plate:” The market for imported and locally-made sterling silver, consumer activism, and national identity in Charleston, 1760–1790, Unpublished dissertation, University of South Carolina, 2009, 31-2.
[6] B. S. Culp, “Mr. Petrie’s ‘Shop on the Bay’,” Antiques and Fine Art (2007), 250-5.
[7] Yale University, Beinecke Library: GEN MSS VOL 184 (folio)
[8] Yale University databases: https://slavery.yale.edu/links/subscription-databases
Slave Makers
Hear Me Now, an exhibition at the Metropolitan Museum of Art, opened this month. A wonderful video allows us to hear the thoughts and reflections of the curators. More importantly, through the art they have curated we are able to hear the voices of the makers, enslaved potters of great skill, put to work at Edgefield in South Carolina. As slaveholders and their enablers tightened laws aimed at restricting literacy, these makers spoke eloquently in local clay, the same kaolin (porcelain clay) that they and their families had worked, or still worked, in West Africa. More than this, they wrote on the pots they threw. Dave, a potter of immense talent, also wrote poetry.
In highlighting the reliance on highly skilled slaves in artisan production, the exhibition reminds us that the antebellum South was far from unique. Slavery has been ubiquitous in human history and we have no way of knowing, in almost every case, whether a work of pre-modern art was produced by an enslaved person.
It was certainly the case in the Roman world that skilled slaves made almost everything, frequently alongside those who had been freed and those who were born free.[i] Private slaves and freedmen were also employed in countless artisanal contexts.[ii]
Enslaved artisans led hard lives, but typically did so in far better conditions than those endured by industrial and agricultural slaves. I highlight here metal communities, since they extracted and smelted the ores to produce metals worked in countless smithies and workshops by artisans. Miners and smelters were frequently imperial slaves, perhaps criminals or prisoners-of-war condemned to their profession, but also those who happened simply to born to miners, and therefore were bound to the mines. A section of the Theodosian Code (CTh.), a compilation of laws produced in the 430s, entitled De metallis et metallariis, addresses the regulation of mines and miners, metals and metal-workers, and stone quarries. A law of 424 (CTh. 10.19.15) ruled that “if any person should be born from a miner and from any other stock, he shall necessarily follow the ignoble birth status of a miner”. Moreover, if miners should “seek to migrate to foreign parts, they shall be recalled to the family stock and the household of their birth status”. There was by then an established body of law relating to the flight of indentured miners, starting with a ruling of 30 April 369, which instructed provincial judges to provide support in identifying and apprehending vagrants (CTh. 10.9.5).[iii]
[i] The vast bibliography can be entered via N. Lenski, ‘Violence and the Roman slave’, in The topography of violence in the Greco-Roman world, eds. W. Riess and G. Fagan (Ann Arbor, 2016), 275-98.
[ii] A related law, issued just a month or two later, suggests miners had absconded to Sardinia by ship, and that ships’ captains were liable for a fine of five solidi for each miner they helped to escaped. Evidently, the practice continued, and a further law of 378 returned to the matter of miners absconding to Sardinia, notably gold miners. There were certainly fewer slaves working the mines than in earlier centuries.
[iii] C. Hawkins, Roman artisans and the urban economy (Cambridge, 2016), passim, especially 84-5, 87, 89, 91-2, 97, 126-8, 130-91.
Ice cores
Just a note on something icy on a hot summer day. As we wrote in New Rome, in a section excerpted in Lapham’s Quarterly, Roman age smelting of silver-lead ore has left signals across the North Atlantic world in the form of anthropogenic heavy metal contamination of soil, sediment, and ice. Cores extracted from glaciers in Greenland and the Arctic show a sudden and dramatic rise in the deposition of atmospheric lead pollutants between c. 100 BCE and CE 100.
Lead is released by the smelting of a range of metallic ores, including those mined for copper and gold, tin, zinc, and silver, and from lead itself. In each location the levels of lead pollutants fall away rapidly toward 400, only beginning to rise again after 800, and not reaching Roman levels until c. 1700. In none of these locations is there any evidence for contemporary mining and smelting of metallic ores, which would have produced the contamination.
Roman-age pollution in the North Atlantic world is the direct result of fluctuations in the intensity of smelting that took place thousands of kilometers to the south, releasing into the atmosphere lead aerosol particles that were conveyed great distances within the northern hemisphere’s atmospheric transport system and deposited by precipitation. The origin of the lead in Greenlandic ice has been confirmed by geochemistry (isotope analysis). Spain was the source of up to 70 percent of the heavy metal pollution at its peak in the first century. Contamination is far greater the closer one gets to its source. In an ice core taken from the Col du Dôme glacier in the French Alps, the magnitude of lead contamination is one hundred times greater than that recorded in Greenland in the first century BCE, reaching a lower peak in c. 100, before falling steadily and dramatically to its lowest point in the sixth century.
In contrast, there are no spikes in lead pollution evident in any of ice cores extracted in Antarctica before around 1890. There is a steady, gradual increase in lead concentrations, reflecting the emergence of metallurgy in the southern hemisphere after CE 1500. Concentrations of lead triple from ~0.6pg g-1 in CE 1650 to ~1.8pg g-1 in CE 1885, then triple again to ~5.4pg g-1 before 1900. The isotopic signature of the lead relates it directly to the commencement of silver-lead mining at Broken Hills, and smelting at Port Pirie, in southern Australia.
According to a team of scientists, principally from the Desert Research Institute:
Concentrations remained high until the late 1920s, with a temporary low during the Great Depression (~1932) and again at the end of WWII (~1948) when concentrations dropped back to mid-19th century levels. Concentrations increased rapidly to 5.7 pg g−1 by 1975 and remained elevated until the mid-1990s. Concentrations during the early 21st century were ~3.7 pg g−1 lower than the peak 20th century concentrations but well above background levels before the start of the Industrial Revolution.(1)
This study draws on sixteen separate cores “from widely spaced coastal and interior sites”. It confirms and expands earlier, similar findings from a study of only the Law Dome glacier.(2)
The contrast between northern and southern hemisphere lead pollution highlights the scale and impact of Roman age smelting.
Notes
(1) J. McConnell et al., ‘Antarctic-wide array of high-resolution ice core records reveal pervasive lead pollution began in 1889 AD persists today’, Scientific Reports 07/2014: 4: 5848, 5 pages.
(2) P. Vellelonga et al., ‘The lead pollution history of Law Dome, Antarctica, from isotopic measurements on ice cores, 1500 AD to 1989 AD’, Earth and Planetary Science Letters 11 (2002), 291-306
Lead pollution, modern and ancient (2 of 2)
Lead has long been used in paint to increase its opacity and improve coverage. However, growing awareness of the dangers of lead has led to the introduction of bans on the production and sale of lead-based paints for domestic use 79 countries, including the USA (1978), and the UK (1992). Not every country has implemented a complete ban, and not every ban has been effectively enforced. China, perhaps the largest international paint producer, has long been suspected of failing to enforce standards, and only very recently introduced tougher standards for the amount of lead allowed in paints (2020).
In 2008, thirty years after lead paint was banned for domestic use in the USA, a study estimated that 70% of elevated blood lead levels (EBLs) identified in children still derived from paint in their homes. 16% of US children (1.7 million) had EBLs, meaning a reading greater than 5.0 μg/dL (micrograms of lead per deciliter of blood). That number has fallen significantly since. Analysis of more than a million blood lead tests conducted between 2018 and 2020 found that fewer than 2% of children had EBLs, although still more than half had detectable blood lead levels.
Children are far more susceptible to lead poisoning than adults. They absorb far more ingested lead from their GI tracts (40% or more, compared to 5-10% by adults) and store it more in soft tissue. The human body mistakes lead for calcium, sending it to places where calcium has vital roles to play. More than 95% of calcium (and therefore of lead) in an adult human body, and 70% in a child, is stored in bones and teeth. Adults excrete lead via the kidneys and liver. Because their kidney and liver detoxification systems are biologically immature, children excrete lead less easily and absorb it more readily into soft tissue and internal organs, including the brain. A child’s blood-brain barrier is also immature and susceptible to damage by lead. Calcium is essential for brain development and function, whereas lead is a powerful “developmental neurotoxin, interfering with neurotransmission, cellular migration, and synaptic plasticity during central nervous system development … [which leads to] many cognitive and motor deficits.”
The same study highlighted the risk of dust: “house dust levels best predict children’s BLLs [blood lead levels]”. Roman children, particularly those in richer households where walls were richly painted, or where paintings might hang, must have been exposed to copious quantities of lead dust.
Roman paint was replete with lead. Analysis of a portrait of a young woman painted on a wooden panel in Egypt (2nd century AD/CE), now held at the National Gallery of Art, Washington D.C., has shown that lead white was used to paint a white necklace and earrings, but also that it was mixed with other substances to paint the woman’s lips (lead white, hematite and charcoal) and skin (lead white, goethite, natrojarosite, hematite), and to provide the painting’s green-yellow background (lead white, natrojarosite, charcoal).
Lead white (lead oxide), known as minium, was produced by immersing lead in strong vinegar (acetic acid). It might then be roasted to produce a red pigment (minium secondarium).
A rich, and very expensive red-orange pigment, vermillion, was produced using cinnabar (Mercury Sulphide, HgS), which like lead white is insoluble in water, so produced excellent washable paint. However, it is also a potent neurotoxin that can be inhaled, ingested or absorbed through the skin. Mercury Sulphide accumulates in the GI tract, liver, spleen and thymus, and may also cause major organ failure.
Some pigments were benign. Perhaps the most commonly used red pigment was produced from hematite (red iron oxide). Hematite is not toxic and was very widely used, for example, in the frescoes of the villa at Boscotrecase (pictured above, from the Metropolitan Museum of Art). However, it, and many other colors, were mixed with lead white to produce lighter shades with better coverage and opacity.
Lead pollution, modern and ancient (1 of 2)
A recent study has demonstrated that lead pollution has damaged the health of half the population of the USA. Lead can be inhaled or ingested. Between 30% and 50% of inhaled lead particles remain in the body, compared to 5-10% of lead particles that are ingested by adults. Lead is equally toxic however it enters the body, and it is especially dangerous for the growing bodies of children, which absorb far more ingested lead (40-50%). For more than a century, lead in paint and later in gasoline has exposed children to a lethal toxin in their homes and streets. (We shall look at lead in paint in another blog post.)
Every person alive today who was born before 1980 has suffered known or unknown effects from breathing the fumes of the gasoline additive tetraethyl lead (TEL). Millions have lived with or died from conditions without knowing that exposure to historical lead pollution may have contributed to them, including cognitive impairment, intellectual disability, and cardiovascular disease.
Despite knowing from soon after its introduction in the 1920s that burning leaded gasoline was deleterious to human health, the fuel additive, which boosts octane and reduces the likelihood of pre-ignition (“knocking“), was only fully banned for regular automobiles in the USA (and Germany) in 1996, following similar bans in other countries. It was only last year, in July 2021, that the last leaded gasoline was used in automobiles in Algeria. Moreover, leaded fuel is still used in aviation and in motorsports. Aviation (the use of “avgas”) emits more than half of the annual lead pollution in the USA, and children who live near airports suffer disproportionately. Although the most popular motor-racing organizations in the USA, NASCAR and ARCA, switched to unleaded fuels in 2007, it remains in common use.
A recent study has suggested that a single three-hour track race can release as much lead into the atmosphere as an average airport does in a year, causing atmospheric lead pollution to jump more than 20% in locations up to 50 miles from the track and persist for up to four weeks. The same study argues that the “social cost” of a burning a gallon of leaded racing fuel, which retails for $10-$20, is $110,000 per gram of TEL added (which varies, and is hard to identify precisely, but appears to be between 4 grams [medium, 110 octane] and 6 grams [high, 116 octane or more] per gallon).
The UK was a laggard in cleaning up its petrol, implementing a full ban only in 2000. Due to lead’s ability to persist in the environment, it has been shown that two decades later “lead deposited via gasoline combustion still contributes significantly to the lead burden in present-day London”. Likewise, in the USA, many studies in smaller and larger urban centers, from Appleton, WI, to Durham, NC, to New Orleans, LA, have shown the persistence of lead pollution in urban soils and argued for necessary remediation.
Cleaning up historical lead contamination, which is expensive and whose costs generally fall on communities and municipalities rather than state and national governments, has rarely been a priority, and therefore historical lead contamination remains a feature of urban environments. The same holds in rural settings.
The use of leaded fuel to run agricultural equipment was phased out less quickly than for regular automobiles in the USA. A presidential report published in 1986 recognized that a great deal of expensive farm equipment would have been damaged (“valve-seat recession“) if run on unleaded fuel. A shift to diesel-powered equipment — diesel was never leaded — took off at that time, but it has not been without significant health consequences for farmers. A further shift to electrification may be slower still because of the high power-to-weight ratio required to run of heavy farm equipment.
Lead from historical pollution remains a minor threat to our soil and food supply. Furthermore, lead (and other heavy metals), which was present in widely used commercial fertilizers, is now less of a problem. However, one must imagine that crops grown near sources of lead pollution, which today include airports and racetracks, remain problematic. Lead fixes in the top 2-5 cm of soil, but regular cultivation will drive it lower into the soil. Unfortunately, this is the root zone for many crops. Lead is drawn from the soil into these crops, and hence is ingested when eaten. There is more to do, therefore, but also an increasing recognition that it must be done even where local resources are inadequate.
Despite the foregoing gloom, there has been amazing progress in removing lead from urban and rural contexts, improving the health prospects of today’s children. Today, children ingest far less lead in their food even as they inhale far less than their parents and grandparents did as children. If lead in water and paint remain issues, modern governments have the knowledge and capacity to regulate and legislate necessary change. University and commercial researchers, large corporations and small businesses have embraced that change and taken it forward through innovation and investment. There are, of course, still local, regional, and national challenges, but the level of environmental lead pollution measured globally has fallen precipitously since reaching a global peak in the 1970s.
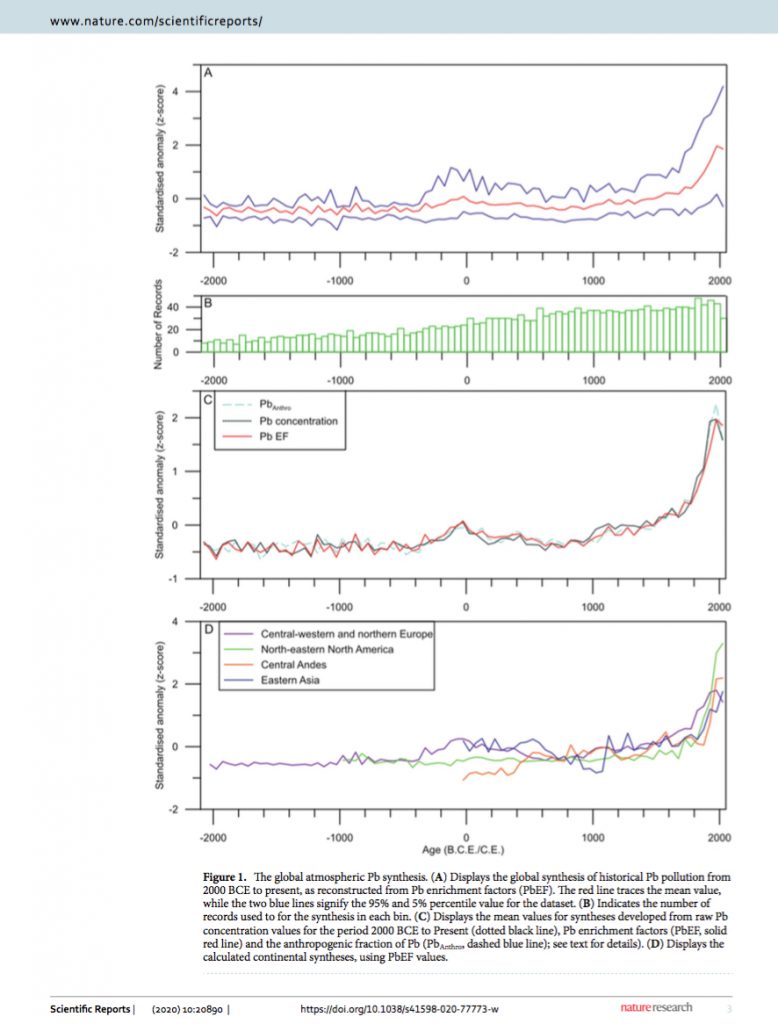
Historical levels of lead pollution are preserved in glaciers and revealed in extracted ice cores. These show that globally lead pollution reached increasingly higher peaks at various points over the past 2000 years. Alas, although falling rapidly, global lead pollution is still at historically high levels. Until the advent of leaded gasoline, each peak was associated with an increase in smelting and metallurgy. Conversely, the only return to something like a “background level” of atmospheric lead pollution has been associated with the demographic and economic catastrophe of the fourteenth century. Ancient Roman lead pollution reached its peak between 100 BC/BCE and CE/AD 100, which period marks the rise and fall of the Roman silver denarius.
Although recycling of lead was common, remediation of lead contamination was unknown in the pre-modern world. Hence, each additional peak in emissions added to environmental lead accumulation, which was far greater the closer one lived to a center for smelting. A good deal of evidence suggests that the Romans did not establish large settlements near mining/smelting facilities. However, recent research in Britain suggest this sensible rule was not universally observed. As yet to be published excavations near Charterhouse-on-Mendip in Somerset, one of the principal lead mining and smelting areas in Britannia, have uncovered a settlement of some size and wealth. It recently featured in a BBC archaeology show, Digging for Britain (season 9, episode 5).
Earlier field surveys at Charterhouse-on-Mendip had identified a substantial settlement, encompassing between 27 and 36 hectares (67-89 acres), which was continuously occupied until the fourth century CE, after which ceramic finds end. For some of the period of occupation it boasted an amphitheatre improvised from an existing earthwork. Clearly, this was not a peripheral mining colony occupied only by imperial slaves supervised by soldiers. It was, however, wholly reliant on imported food and fuel. Most significantly, there is no evidence for local agriculture to support the substantial settlement and related settlements at Priddy and Green Ore. This suggests clearly an awareness of the consequences of smelting on the local environment. [i]
Charterhouse-on-Mendip, where thin seams of galena (lead sulphide ore) exist near the surface of a limestone landscape, sits above a complex cave system. Mining is known at this site from pre-Roman times, and Roman slags contain up to 25% lead, more than a third of the original lead content before smelting. Whereas unprocessed galena is not soluble in water, roasted ore slag is more soluble and atmospheric fallout from smelting is highly soluble. Water permeating through the surface limestone into the caves has left clear indications of surface contamination in speleothems, mineral cave deposits formed from dripping water. One speleothem, in this case a stalagmite, suggests a peak in lead contaminants between CE 50 and 150, followed by a further and higher peak between CE 400 and 500, which falls away rapidly afterwards. This may relate to leaching from slag deposits after smelting activity had ended. However, it is also likely that mining and smelting continued in the Mendips for far longer than elsewhere, since it was known that these mines yielded the most silver. This is consistent with the continued but far lower levels of pollutants after CE 200 at bogs hundreds of miles to the north of the Mendips. There is no evidence for medieval reoccupation of the settlement at Charterhouse, which remained contaminated, but there was medieval mining activity. [ii]
If the most contaminated sites in Roman Britain were not used for arable or pastoral farming, still substantially increased levels of lead aerosols deposited across a broad region by precipitation will have affected bees and birds, fish and shellfish in polluted rivers and bays, cattle and sheep eating grass growing in contaminated soil. Lead entered the food chain and affected human health across northern Europe, hundreds of miles from smelting facilities. Consequently, many Roman children will have suffered in similar ways to modern children exposed to elevated levels of lead pollution and no efforts were made to remediate the environmental damage done by Roman-age smelting.
[i] M. Fradley, ‘The field archaeology of the Romano-British settlement at Charterhouse-on-Mendip’, Britannia 40 (2009), 99-122.
[ii] D. McFarlane t al., ‘A speleothem record of early British and Roman mining at Charterhouse, Mendip, England’, Archaeometry 56 (2013), 431-43.
Structural violence
Violence is commonly understood to comprise actions that threaten or cause injury or death. This works well as a succinct definition of ‘direct violence’, which includes suicide, homicide, mass killings, child abuse, terrorism, rape, domestic abuse, etc. Most of these categories can be identified in the Roman world, appearing in a range of texts. We might begin by exploring the terms used to describe violence and violent acts, in Greek βία and in Latin vis, which can both also mean bodily strength and force, and proceed to their antitheses, the broad categories of peace (εἰρήνη) and love (ἀγάπη), which were foundational tenets of political-religious institutions that both mitigated and justified violence. One must accumulate and analyze abundant empirical evidence, such as anecdotes from ancient biographies and histories, saints’ lives and chronicles, or spells and remedies from magical and medical treatises. One might then attempt to estimate the valorization of violence in the construction of gender, the social experience and effects of direct violence, such as crime in a town or village, and concurrently explore violent sanctions and punishments enshrined in law, for example the healing function of legal punishments (ἐπιτίμια ἰατρευτικά), what we might today call rehabilitation, rather than their punitive quality per se (κολαστικά). A recent study of conjugal violence has highlighted a circle of violent actions, whereby violence by men inspired spiritual or magical violence by women, casting spells on men to cause impotence, which occasioned a physically violent response. These are fascinating themes, but they comprise only a segment of the domain of violence in the Roman world, which is largely lost to us.[i]
Approaching violence through the material record, one might choose to start with evidence for direct violence caused by tools and weapons. Of course weapons were made and used throughout the Roman period in all regions to injure and kill, and this can be seen in a range of written records, in art and the visual record, in the survival of weapons themselves and armor to protect against weapons, and in the archaeological record, including the bioarchaeological record. Injuries caused by sharp or pointed weapons, by swords and spears, but also lead shot for example, can be identified and in cemetery contexts quantified.
An accumulation of violent acts built from the bottom up, of wounds that maimed or killed, of things that caused those wounds, will always be incomplete. It may be representative of direct violence, but will comprises only a fraction of the ‘structural violence’ in a given society, meaning the conditions that result in the premature deaths of or injury to its people. These conditions derive from the social, political and economic structures that define a culture, and the environmental factors by which these are constrained and sustained.
Histria on the Black Sea coast, close to Rome’s lower Danuber frontier, offers insights and illustrations from one modest settlement. Skeletons exhumed from the city’s late Roman graveyard displayed signs of hard, repetitive labour, poor diet and personal hygiene, infection, trauma and chronic pain. A young woman, perhaps as young as 24, was buried with an infant who died at around three months. Despite her youth, the woman displayed signs of severe joint degeneration, and appears to have died with, and perhaps as the result of, a wound to her right leg. She had earlier suffered a wound to her pelvis and a blow to the head with a blunt object, both healed without signs of infection. Another woman, buried in an adjacent grave, died between the ages of 33 and 46. At the time she would have been suffering from both acute and chronic pain. She had lost a tooth, and had dental caries in two more and four abscesses. Her remaining teeth, molars, canines and incisors, were all heavily or abnormally worn. Her back and limbs showed signs of osteoarthritis and degeneration, and her right hand would have appeared deformed from damage and wear. She would have walked hunched over, although once stood 1.59m (5’2”) tall. The man buried next to her was around the same age at death and 1.71m (5’7”) tall. He had lost ten teeth and had caries in four more. He suffered from severe osteoarthritis and eleven of his vertebrae were fused.[ii]
We might then consider environmental violence, the violence done to the natural world, to flora and fauna, including humans, by human behavior and activities. Clearly, this is a vast topic and one that demands circumscribed points of entry and paths through massive bodies of data, written, material and scientific. Devastating Beauty will adopt works of art as those points of entry.
[ii] C. Radu et al., ‘Bioanthropological data for a skeletal sample retrieved from the extra-mural necropolises at Histria’), in Histria: histoire et archéologie en Mer Noire, Pontica 47, supplementum III (Constanța, 2014), pp. 283-97.
A late Roman lead coffin
A lead coffin (Met 61.206), at only 36cm long, would have held a newborn infant from Tyre, today in Lebanon. Now on display at the Metropolitan Museum of Art, a coffin of this size is rare and speaks to the modest wealth of the infant’s parents, to their desire to inter their child with proper ceremony. It bears witness to parental love and the hope for salvation, the skills of a local craftsman, and the chthonic qualities of lead, re-deposited in the earth thousands of miles from where it was extracted. The coffin’s decoration was formed by pressing designs into a mold of clay or sand into which the molten lead was poured. The designs betray their workshop, and each mold was unique. On the lid of the infant’s coffin are two knotted, braided ropes and on the sides, ends and lid are imprinted rosettes and columns, apotropaic symbols. The ropes figuratively bind the coffin and keep it closed, protecting the body from malign spirits. The coffin speaks to the fragility of human life in a society where life expectancy at birth was always below 30 years. Some were poisoned by lead, which the young absorb more readily than adults. Extant works of art in lead are rare, in part because lead is so easily recycled, and in part because it was not regarded as an appropriate medium for art. Lead was regarded as the antithesis of silver, a gleaming noble metal extracted from the same ore, lead sulphide (galena).
The lead used to form the infant’s coffin almost certainly came from Britain or Spain, the empire’s two principal sources through the fourth century. It is remarkable that a tradition of burial in lead developed in later Roman Syria, far from any natural sources of lead. The rarity of lead coffins from Roman Spain suggests no such tradition had developed there, where lead was abundant. Only, in Britain, where lead was also plentiful, does one find a tradition to rival or surpass that of Syria.
Of the now more than 300 lead coffins known from Roman Britain, around two-thirds have been dated to the fourth century. The distribution of finds suggests the principal determinant of whether one was buried in lead was wealth, but a large number of coffins near the Mendips indicates that the availability of lead and of lead workers was also a factor. One might suggest that the availability of lead in Britain led to the popularity of lead coffins in an age of inhumation, but would require a quite different explanation for their popularity in the Levant, where all lead was imported. Moreover, cremated remains have also been found inside lead containers, for example a lead-lined stone coffin at Harnhill, Gloucestershire.[i]
Spitalfields woman, a fourth-century resident of Roman London, was buried in a coffin of British lead within a stone sarcophagus. The lead of her coffin was quite different from that in her tooth enamel, and which stained her bones brown. Strontium and lead isotope analysis of her tooth enamel has determined that she is very likely to have grown up in the city of Rome. The concentration of lead detected in her tooth enamel was sufficient to indicate lead poisoning in childhood, and the isotopic signature was different to that of native Britons. The woman’s lead intake was sustained, producing still greater concentrations in adulthood. A black gingival line around her teeth suggests this woman suffered also from lead poisoning as a young adult, surely through regular ingestion with food and wine.

Spitalfields woman’s lead coffin was decorated with a design formed by pressing scallop shells into the sand in which the lead sheets were formed. Braided rope patterns appear to bid the lid to the coffin, in a manner similar (but not identical) to the coffin of the Tyrian infant. The shells perhaps allude to paradise, although in Britain’s coastal waters at this time scallops will have ingested a great deal of lead contamination.
Another young immigrant woman who died at around this time was buried at York, also in a stone sarcophagus (already broken upon its discovery in 1901), although no lead liner was recorded. ‘Ivory Bangle Lady‘ had emigrated from North Africa in the fourth century. A carved ivory plaque discovered with her skeleton, a gift from her sister in life or in Christ, reads ‘SOROR AVE VIVAS IN DEO’, ‘Hail sister, may you live in God’. It is possible that the plaque was given as a baptismal gift and later, not necessary much earlier than she died. It was but one of several grave goods, including jet bangles, a silver pendant, glass earrings and beads, and a mirror. VIVAS is the most common word in late Roman inscriptions on objects found in Britain. It conveys hopes for a happy life and the best wishes of a friend of family member, frequently in a Christian context. VIVAS appears on at least seventeen silver spoons dated to the third and fourth centuries discovered in Britain at Chedworth Villa, Colchester, Dorchester, Great Horwood, Mildenhall, St Neots, and Thetford. On each occasion, it accompanies a name, the person being wished ‘life’. These have been interpreted by some as christening spoons, gifts given upon baptism, hence wishing ‘eternal life’ to the recipient.[ii]
VIVAS also appears on some rare fourth- or fifth-century lead caskets from Britain. The Caistor Casket, discovered at Caistor near Lincoln, was discovered in 1863 as three separate side panels ‘in a crumpled mass, weighing over 50 lb.’, when a drain was being dug under the road west of the churchyard. One of these pieces ended up in the Lincoln Museum, today The Collection, Lincoln, while two were deposited at the British Museum.[iii]
Notes
[i] N. Wright, ‘A lead-lined stone coffin cremation burial from Harnhill, Gloucestershire’, Transactions of the Bristol and Gloucestershire Archaeological Society 126 (2008), 83-90; B. Barber et al., ‘Recent excavations of a cemetery of Londinium’, Britannia 21 (1990), 1-12.
[ii] J. Eisinger, ‘Lead and wine. Eberhard Gockel and the colica Pictonum’, Medical History 26 (1982), 279-302; H. Shaw et al., ‘Identifying migrants in Roman London using lead and strontium stable isotopes’, Journal of Archaeological Science 66 (2016), 57-68. Adults store up to 94% of absorbed lead in their bones and teeth, children only 70%, making it more toxic to soft tissue in the young.
[iii] C. Hawkes, I. Richmond and V. Nash-Williams, ‘The archaeology of Lincolnshire and Lincoln: the Roman occupation’, Archaeological Journal 103 (1946), 16-84, 23-5. There is no evidence for a ritual or votive deposition in a well or watery site in the manner advanced as typical by Petts 2003.
Lead Production in Roman Britain
Roman lead production began immediately after the conquest of Britain in AD 43; the first marked ingot to have survived bears the year AD 49. The principal lead mines in Britain were in Somerset (Mendips), Shropshire, Flintshire in northern Wales, south Yorkshire (north Pennines) and Derbyshire (south Pennines).[i] There is evidence for Iron Age lead extraction and production, prior to the Roman conquest, at all of these locations, generally for the manufacture of everyday objects like spindle whorls and fishing weights, but also rings and bracelets.[ii]

Tacitus (Agricola 12) considered the acquisition of ‘gold and silver and other metals as the prize for the conquest’ of Britain. However, British ores had very little silver. Ores from the Yorkshire and Derbyshire contain the least amount and that from the Mendips the highest by far. Yet Britain’s ores were so rich in lead that only about 75 percent of the available lead was regularly extracted from galena.[iii] Writing around three decades after the Roman conquest of Britain, Pliny the Elder commented, in his Natural History, 34.49, ‘in Britannia it [lead] is found in the upper stratum of the earth, in such abundance, that a law has been spontaneously made, prohibiting any one from working more than a certain quantity of it’.[iv]
For more than a century after the Roman conquest of the island, lead ingots produced in Britannia, called ‘pigs’, were marked with an indication of weight, where and when they were produced, frequently with the names of emperors, starting with Claudius until Marcus Aurelius.[v] More than 100 pigs are now known, and new finds are made occasionally, either near known production sites or along Roman transportation routes. It seems likely that some pigs were stolen during transit and well hidden, hence they have survived untouched to be found today. Early pigs explicitly indicate military control of the mines. Inscriptions disappear from British pigs, as they do elsewhere in the empire, after Marcus Aurelius (d. 180).[vi] This has been interpreted as an indication of decreased imperial control and lower levels of lead production from the start of the third century, although unmarked pigs continued to be found, several in fourth-century contexts.[vii] In a short but important article, Vincent Gardiner posited a military phase of production, followed by an early commercial phase (late first – early second century, when companies, societates, controlled production), a Hadrianic phase (when huge amounts of lead were required for construction), and a later commercial phase from the later second century.[viii]

Gardiner also provided analysis of the distribution of lead pigs, showing clearly that they are concentrated as one would expect within a zone around the site of production, close to navigable rivers (37%) or Roman roads (24%), with a greater spread to the side of the point of origin that approaches a port from which the ingots might be exported.[ix] Lead from the Mendips appears to have been exported from the port of Clausentum, modern Bitterne near Southampton, on England’s south coast. For the mines of Yorkshire and Derbyshire, the principal port from which pigs were exported was Petuaria (Brough-on-Humber), on the east coast. Eight pigs discovered at Brough-on-Humber are marked as products of Lutudarum,[x] a mine to the north of Derby, in the east Midlands. Additionally, these pigs all bear the mark BR or BRIT, presumably for BRITANNICUM, British, which strongly suggests they were marked for export. Six pigs from the same mould have been identified, all cast to an approximate 200-libra standard and weighing between 184 and 196lbs (except one damaged ingot, RIB 2404.47, which has lost part of its back and right end, weighing 135lbs). They are inscribed G[AI] I[ULI] PROTI BRIT[ANNICUM] LUT[UDARENSE] EX ARG[ENTARIIS], which might be translated as ‘[Produced by] Gaius Julius Protus: British [lead from] Lutudarum, from the silver[-lead works]’.[xi]
We do not how far afield British lead was exported, but lead isotope analysis is beginning to offer compelling insights, for example isotope signatures of British lead have been identified in lead pipes discovered in Pompeii and at Rome itself. Lead coffins produced in Syria surely contained British lead. Shipwreck evidence has long provided clues, including off the coast of Syria. A notable, but vaguely dated wreck, probably of the third or fourth century AD, off the coast of Brittany saw 271 lead pigs spill onto the sea floor. These can only have been from Britain. Analysis of copper alloy jewelry excavated from graves in Kissi, northeastern Burkina Faso, demonstrate that lead from Britain, Spain and Sardinia had made its way via Roman north Africa to sub-Saharan west Africa, surely within metal objects melted down and remade locally. We believe West African gold was travelling in the opposite direction.[xii]
Notes
[i] On different ages and natures of formation, see S. Moorbath, ‘Lead isotope abundance studies on mineral occurrences in the British isles and their geological significance’, Philosophical Transactions of the Royal Society of London, Series A, Mathematical and Physical Sciences 254, no. 1042 (1962), 295-360.
[ii] M. Ponting, ‘Pretia Victoriae or just an occasional bonus? Analysis of iron age lead artefacts from the Somerset lake villages’, Oxford Journal of Archaeology 37 (2018), 185-99.
[iii] This was not for lack of ability. See R. F. Tylecote, ‘Roman lead working in Britain’, British Journal for the History of Science 2 (1964), 25-43.
[iv] Pliny also made other intriguing comments that have vexed interpreters, for example referring to ‘white lead’ (plumbum album) as well as ‘black lead’ (plumbum nigrum). While it is clear that ‘black lead’ is lead, ‘white lead’ has been understood both as silvered lead (i.e. the metal prior to cupellation) and tin. It seems clear to me that the latter interpretation is correct, as it makes greater sense of Pliny’s slightly later observation, in the same chapter, that an alloy called argentarium is made by mixing together black and white lead which is used for soldering pipes. Plumbum nigrum, if it is tin, in Pliny’s day in Rome, cost 80 denarii per pound, argentarium (pewter, with a ratio of 66% tin and 33% lead) 70 denarii per pound, and black lead only 7 denarii per pound. The price difference between is interesting, and one must imagine it held so long as tin remained more scarce than lead, and that both lead and tin were far less expensive where they were naturally abundant, for example in southwestern Britain. There were productive tin mines in Cornwall, close to the Mendip lead mines in Somerset, but not elsewhere in Britain.
[v] Roman Inscriptions of Britain, II.1, ed. S. Frere, M. Roxan and R. Tomlin, 38-46, ‘RIB 2404: Lead pigs’.
[vi] RIB II.1, 65, no. 2404.72a. A pig, probably of British origin, has been discovered in Bourgogne, France, that is inscribed as the property of Septimius Severus
[vii] RIB II.1, 38.
[viii] V. Gardiner, ‘An analysis of Romano-British lead pigs’, Institute of Archaeo-Metallurgical Studies 21 (2001), 11-13. This may be compared with the Spanish situation, for example as interpreted by B. Díaz Ariño and J. A. Antolinos Marín, ‘The organization of mining and metal production in Carthago Nova between the late Republic and early Empire’, Athenaeum 101/2 (2013), 535-53, which posits an early proliferation of small mining companies, from the second to first centuries BC, followed by a period of consolidation and the domination of a few large companies (societates) until the middle of the first century AD, when the mines closed.
[ix] Gardiner 2001, 13; RIB II.1, 38, suggests that the discovery of some lead pigs in pairs may suggest transportation by packhorse.
[x] A. Shone, ‘The search for Lutudarum, evidence and supposition’, Derbyshire Miscellany 17 (2006), 106-16, sets out a strong case that Lutudarum must be identified with Wirksworth, despite little material evidence. A. Breeze, ‘The name of Lutudarum, Derbyshire’, Britannia 33 (2002), 266-8, prefers Carsington, on the basis that excavations have taken place there. See also RIB II.1, p. 56.
[xi] A. Bradwell, ‘Derbyshire Roman lead pigs and “Lutudarum”’, Mining History: Bulletin of the Peak District Mine Historical Society 19 (2014), 14-16; RIB II.1, 56-61. However, the meaning of EX ARG. has been discussed further, notably at Gardiner 2001, 12. Bradwell 2014, has suggested EX ARG refers to the assaying of the ingots to determine that they are ‘without silver’, meaning insufficient to warrant cupellation (removal of silver). The Lutudarum pigs do have very small amounts of silver, but Derbyshire lead sulphide contained very little silver. Two of the pigs from Petuaria also have fragments of lead sulphide that have survived smelting, demonstrating that cupellation has not taken place. Analysis of the three lead vessels in The Collection at Lincoln failed to detect any silver. This is not unusual. Around three-quarters of lead ingots discovered in Britain have levels of silver that are undetectable or below 0.01%/120 ppm.
[xii] H. Delile et al. ‘Lead in ancient Rome’s city waters’, fig. S3; H. Delile et al. ‘A lead isotope perspective on urban development in ancient Naples’, PNAS 113 (2016), 6148-53. See also H. Delile et al., ‘Rome’s urban history inferred from PB-contaminated waters trapped in its ancient harbor basins’, PNAS early edition (August 2017); T. R. Fenn et al., ‘Contacts between West Africa and Roman North Africa: archaeometallurgical results from Kissi, Northeastern Burkina Faso’, in Crossroads / Carrefour Sahel. Cultural and technological developments in first millennium BC / AD West Africa. Développements culturels et téchnologiques pendant le premier millénaire BC / AD dans l’Afrique de l’Ouest, eds S. Magnavita et al. (Frankfurt, 2009), pp. 119-46; T. Garrard, ‘Myth and metrology: the early trans-Saharan gold trade’, Journal of African History 23 (1982), 443-61; A. Wilson, ‘Cyrenaica and the late antique economy’, Ancient West and East 3 (2004), 143-54.
Roman lead pollution
A short excerpt from the curator’s new book has been published at Lapham’s Quarterly as Ancient Roman Pollution.








